1 Introduction
The popular view of creativity is that it opposes rationality and control. Sometimes this is put in terms of hemispheric dominance: those who are rational and controlled are called “left brained,” while those who are creative and impulsive are called “right brained.” Other times it is put in terms of inhibition: creative people are “freer” or less inhibited than other people. Scientific accounts of creativity sometimes lend credence to this popular view (see, e.g., Kaufman et al., 2010, p. 217). We take a different tack: we see control as playing a key role in creative cognition, which we explore through the mechanisms of attention. As we will argue, not only is attention an essential part of the creative process, the more varied and dynamic the uses of attention, the more creative the agent is able to be. Our account draws on evidence from psychology and neuroscience to offer a perspective that is distinctive, while also building on and synthesizing other recent accounts.
As is noted in most other accounts, creativity is a process that aims for novel ideas or products that are useful to some question or enterprise (Runco & Jaeger, 2012). This process can be divided into two phases: idea generation and idea evaluation. In the first phase multiple ideas are generated, whereas in the second phase the most appropriate ideas are selected and further elaborated (Allen & Thomas, 2011). What is often overlooked is that selection is at the core of both phases. In the first phase, selection is used to bring forward the appropriate elements, such as words, musical notes, or shapes, which can then be combined to generate ideas. In the second phase selection is used to evaluate the ideas, separating the good from the bad. Further, this type of selection counts as attention. That is, while selection is possible without attention, task-related selection is only possible with attention. The type of selection required for both phases is task-related selection (albeit sometimes with a task that is widely drawn). Thus, to understand creative cognition, researchers must explore the role of attention.
In this paper we argue that creativity is increased in the dynamic use of multiple forms of attention, rather than in its absence. Two major taxonomies are discussed: top-down versus bottom-up, which is based on the source of attention (Connor et al., 2004; Posner, 1980; Treisman & Gelade, 1980), and internal versus external, which is based on its target (Chun et al., 2011). The studies we review yield inconsistent results regarding the contributions of each taxon to creative cognition. While some find reduced top-down attention to facilitate creative cognition, at least for some tasks (Chrysikou et al., 2013; Reverberi et al., 2005), others report that increased top-down attention is conducive to creativity (Nusbaum & Silvia, 2011; Silvia, 2015). Many studies assign a significant role to internal attention but diverge on possible contributions of external attention. Our account helps to explain these seeming contradictions.
Here, we use behavioral and neuroimaging studies to argue against an either/or narrative when it comes to attention and creative cognition. Instead, we argue that creative cognition benefits from a dynamic interplay between typically opposed attentional mechanisms—top-down and bottom-up as well as internal and external. That is, whereas bottom-up and top-down attention are typically seen as in conflict, as when a distracting sound grabs your attention away from your current task, in creative cognition they work together. Similarly, whereas internal and external attention normally seem opposed to one another, as when rehearsing a conversation prevents one from noticing something nearby, creative cognition is improved by cross-engagement. As we will show, interplay between these forms of attention is reflected in dynamic interactions across three large-scale brain networks: the default mode (DMN), frontoparietal control (FPN), and salience (SN) networks. In what follows we explain the unique role of attention in creative cognition and the contributions of each type of attention while supporting our claim that a dynamic relationship between multiple attentional mechanisms best supports creativity. Consequently, we propose a two-dimensional space of attention that sufficiently captures the flexibility of creative cognition and unifies seemingly contradictory accounts of its underlying attentional mechanisms. Along the way we will also provide background on the dual-process account of creative cognition, the major taxonomies of attention, and how these are related.
2 Background and motivation
Major accounts of creative cognition depict it as a multi-phase phenomenon, including an explorative phase wherein many ideas are generated and an evaluative phase where ideas are further assessed and elaborated (Allen & Thomas, 2011; Runco & Basadur, 1993). For example, Wallas (1926) proposed a four-stage model of creative problem solving: preparation, incubation, illumination, and verification. Individuals confront the problem in the preparation phase, stop consciously thinking about the problem in the incubation phase, experience an “aha” moment when they find a solution in the illumination phase, and then actively test the solution in the final verification stage. Wallas (1926) suggested that only the first and last stages require reasoning, whereas the middle stages instead depend on imagination.
Streamlining these ideas, Guilford (1950, 1967) proposed a dichotomy of idea production strategies, known as divergent and convergent thinking. Divergent thinking is the ability to generate many ideas and is considered most useful for exploration and solving open-ended problems. Convergent thinking is thought to be more analytical and suited for problems with one correct answer. Guilford (1967) claimed that divergent thinking plays a more significant role in creativity, but that convergent thinking also contributes. Divergent thinking ability is often assessed through tasks such as the Alternative Uses Test (AUT, Torrance, 1966), in which the participant is asked to come up with many alternative ways of using a simple object, such as a paperclip, whereas convergent thinking ability is commonly evaluated through the Remote Associates Test (RAT, Mednick & Mednick, 1967), in which participants are asked to identify a word related to three cue words, such as “cheese” for the cue words “cottage,” “swiss,” and “blue.”
Campbell (1960) applied principles from evolutionary theory to propose a different set of phases. He contended that creative thinking comprises two stages: random variation and selective retention. In biological evolution, variation is produced through random mutations and the recombination of old genes.1 New variations are not intentionally designed to enhance adaptation, but the gene combinations conducive to species survival are more likely to pass on to future generations. Relatedly, Campbell described idea generation as a process that allows for explorations “beyond the limits of foresight or prescience” (Campbell, 1960, p. 381). Once the generated ideas fit some criteria—set by intrinsic or environmental constraints—they are selected and stored in memory to produce further variations (see also Simonton, 1999).
Aligning with these multi-phase descriptions of creativity, neuroimaging studies have rendered supporting evidence for a “biphasic” or “dual-process” account of creative cognition (Beaty et al., 2018; Ellamil et al., 2012; Liu et al., 2012; Rosen et al., 2020). Such studies concur that creativity starts with an initial stage of idea generation followed by the search for an optimal solution through idea evaluation. The overwhelming evidence from such studies is that there is no specialized brain area or mechanism dedicated to creative cognition. Instead, creativity emerges from dynamic interactions between common cognitive processes, including executive function, memory, and attention (Abraham, 2018).
What is missing from the above accounts is a concrete description of the cognitive mechanisms underlying each phase of the creative process and how those mechanisms are implemented in the brain. In this paper we take some initial steps toward remedying this gap by focusing on the role of attentional mechanisms in creative cognition in both behavioral and neural terms. Future work will be required to develop this account into a testable model.
3 The role of attention in creative cognition
The ubiquity of attention in contemporary psychology and neuroscience is paired with a lack of agreement as to its definition. It is a running joke that James once stated: “everyone knows what attention is” (James, 1890, p. 403), while others assert that “no one knows what attention is” (Hommel et al., 2019). Perhaps the most agreed upon feature of attention is that it concerns selection: the prioritization of some mental events or neural processes over others. Beyond that, there is considerable disagreement as to its unity and scope. Yet, most would agree that attention plays a central role in cognition; it was, after all, at the heart of the so-called “cognitive revolution” (Nobre & Kastner, 2014). We agree with Nobre and Kastner that the “consensus definition” is something like “the prioritization of processing information relevant to current task goals” (Nobre & Kastner, 2014, p. 1214).2 Because this prioritization can happen at different levels of analysis and in many different parts of the brain, we discuss below two specific taxonomies of attention as they relate to creative cognition: top-down versus bottom-up and internal versus external attention.
3.1 Top-down versus bottom-up attention
The difference between top-down and bottom-up attention is based on the source of input to the process of prioritization: top-down inputs include all those that are related to the current task (e.g., one’s motivation to focus on the task) whereas bottom-up inputs include all those that are unrelated to the current task (e.g., salient stimuli that might distract from the task). For example, Buschman and Miller (2007, p. 1860) write: “Attention can be focused volitionally by ‘top-down’ signals derived from task demands and automatically by ‘bottom-up’ signals from salient stimuli.” Top-down attention is thus sometimes referred to as “endogenous” or goal-directed, whereas bottom-up attention is referred to as “exogenous” or stimulus-driven (Posner, 1980).
In neural terms, top-down and bottom-up attention correspond with different systems of activation (see, e.g., Jennings, 2020, for an overview). Bottom-up input is feedforward starting from sensory cortices, whereas top-down input relies on feedback from higher-order neural areas (see, e.g., Katsuki & Constantinidis, 2014 for an overview). Specifically, top-down attention operates through the frontoparietal network (FPN), which is responsible for executive control in the brain. The FPN primarily comprises the dorsolateral prefrontal cortex (dlPFC) and posterior parietal cortex (PPC) and contributes to executive functions such as task initiation, task maintenance, and task switching (Cole & Schneider, 2007; Vincent et al., 2008). Because it relies on feedback from higher-level neural areas, top-down attention operates about 50-60 milliseconds slower than bottom-up attention (Baluch & Itti, 2011; Corbetta & Shulman, 2002).
While these forms of attention can be examined as separate processes (e.g., Treisman & Gelade, 1980), they can also be examined as interacting forces in overall resource distribution (e.g., Connor et al., 2004). This is sometimes described through the language of a “priority map”: “correlates of bottom-up and top-down attention reveal coactivation of the same network of parietal and prefrontal cortical areas, giving credence to the idea of a priority map simultaneously modulated by both types of factors” (Katsuki & Constantinidis, 2014, p. 510). In what follows we assume that top-down and bottom-up input typically interact in order to guide attention, rather than thinking of them as entirely separate processes, with “top-down attention” reflecting greater priority given to top-down input, and “bottom-up attention” reflecting greater priority given to bottom-up input.
The role played by top-down and bottom-up attention in creative cognition has been a point of contention (Dietrich & Kanso, 2010). While some accounts claim that reduced top-down attention supports creative cognition, others claim the opposite: that increased top-down attention brings about creativity (see below). In this section we argue that these accounts are compatible when we consider the different phases associated with creative cognition, managed by the agent through her control of attention.
3.1.1 Reduced top-down attention as key to creative cognition
On one account, reduced top-down attention is conducive to creative cognition (for reviews, see Chrysikou, 2018; Zabelina, 2018). According to this account, reduced top-down attention, marked by deactivations of dlPFC, facilitates spontaneous associations and the generation of novel ideas (Carson et al., 2003; Kasof, 1997).
As evidence for this account, it is reported that spontaneous jazz improvisation is characterized by deactivation of areas of the prefrontal cortex (Limb & Braun, 2008). It is also reported that patients with damaged PFC exhibit better performance on creative tasks such as matchstick problems (Reverberi et al., 2005) and drawing (Shamay-Tsoory et al., 2011). In addition, reducing prefrontal activity in healthy subjects—through noninvasive transcranial direct current stimulation—leads to better creative performance in tasks such as AUT (Chrysikou et al., 2013). Similarly, lesions in PFC are associated with improved performance in creative tasks that require overcoming the constraining influence of recently activated knowledge (Abraham, 2018). Along those lines, children younger than five years old encounter less functional fixedness—the tendency to perceive an object only in terms of its most common use—which is consistent with reduced top-down attention given the late development of PFC in children (German & Defeyter, 2000; Gopnik et al., 2017).
Reduced top-down attention allows for more openness to bottom-up stimuli that might not be directly relevant to the task at hand. For example, studies report that creative individuals are more sensitive to peripheral auditory cues (Ansburg & Hill, 2003). Increased sensitivity to bottom-up stimuli is itself hypothesized to help creativity in two ways: 1) these stimuli might lead someone to make new connections between remote concepts, and 2) these stimuli expand an individual’s repertoire of potential building blocks for future creative ideas (Zabelina, 2018).
3.1.2 Increased top-down attention as key to creative cognition
In contrast, other research finds that increased top-down attention promotes creative cognition. Top-down attention, for instance, plays an important role in guiding memory retrieval, as well as in identifying and deploying task strategies, all of which support creative cognition. Further, top-down attention is necessary for inhibiting habitual responses that would otherwise impede creative performance (Aziz‐Zadeh et al., 2009; Bengtsson et al., 2007; Edl et al., 2014).
Evidence supporting this approach includes studies that report a positive relationship between creativity and inhibition. For example, Edl et al. (2014) used a modified Stroop task to measure cognitive control. The Stroop task is a standard clinical and experimental measure of inhibitory control that asks participants to name the color a word is printed in, rather than the printed word, which requires them to inhibit the automatic tendency to read the printed word (see, e.g., Herd et al., 2006, p. 79). They found that creative individuals did not experience Stroop interference, whereas the control group exhibited a robust interference effect.3 The researchers concluded that more creative individuals “show stronger cognitive control” and “more efficiently suppress competing dominant but irrelevant actions, processes, or mental activities” (Edl et al., 2014, p. 41). This research was supported by a follow up study that found “inhibition significantly predicted creativity (i.e., divergent thinking ability)” (Benedek et al., 2014, p. 79). In another study, Bengtsson et al. (2007) reported increased activation of cognitive control areas (i.e., dlPFC) in the generation of musical structures during improvisation. They concluded that cognitive control is necessary to adapt the improvisation to an “overall goal of producing an aesthetically satisfactory end-result” (Bengtsson et al., 2007, p. 837).
3.1.3 Flexible interactions between top-down and bottom-up attention as key to creative cognition
The accounts we reviewed above seem contradictory. Some attribute creative cognition to reduced top-down attention, arguing that it facilitates unconventional, spontaneous thinking. Others consider increased top-down attention as conducive to creative cognition, arguing that top-down control is necessary for goal-directedness and the inhibition of conventional ideas in favor of novel ones. Both accounts are supported by numerous behavioral and neuroimaging studies (see above).
To address these seemingly inconsistent findings, Pinho et al. (2015) conducted an experiment using functional magnetic resonance imaging (fMRI). They instructed professional pianists to perform improvisations on a piano keyboard using either specific keys (“key condition”) or specific emotional content (“emotion condition”). Pinho et al. found that the key condition corresponded with higher activity in dlPFC and greater functional connectivity between dlPFC and premotor and parietal areas.4 In contrast, in the emotion condition dlPFC was less active overall but had greater functional connectivity with a neural network known as the default mode network (DMN; this network will be further explained below). Thus, Pinho and colleagues concluded that activation of dlPFC is not intrinsic to creative cognition:
Creative performance can be associated with both an increase and reduction of activity in the DLPFC (and other working memory regions) within-person, depending on task prerequisites. The findings clearly indicate that creativity is the result of a dynamic interplay between several brain regions, networks, and systems, and that the patterns of brain activity during creative problem solving depend strongly on the employed problem solving strategies. (Pinho et al., 2015, p. 3061)
This study suggests that instead of focusing on a specific form of attention as the key to creativity, one should focus on the agent’s flexible use of multiple forms to support the task at hand.
Multiple meta-analyses support this approach to creative cognition, using a coordinate-based method to detect overlaps between brain areas reported by individual studies (Boccia et al., 2015; Gonen-Yaacovi et al., 2013). These meta-analyses indicate that creative cognition is supported by distributed neural networks comprising brain areas associated with both top-down and bottom-up attention. For example, Gonen-Yaacovi et al. (2013) reported that many creative activities share common neural correlates, including areas associated with both top-down (e.g., lateral prefrontal cortex) and bottom-up attention (e.g., angular gyrus), regardless of domain (e.g., verbal, musical, visual). Yet, each creative activity also has its own neural signature. For example, activation of motor planning areas is more strongly associated with musical improvisation tasks relative to verbal tasks (Boccia et al., 2015).
These results suggest that creative cognition is supported by a flexible interplay between top-down and bottom-up attention. This flexible interplay corresponds with the phases of creative cognition: reduced top-down and increased bottom-up attention benefits the idea generation phase of creative cognition, while increased top-down attention benefits the evaluation phase (Beaty et al., 2016; Ellamil et al., 2012; Kleinmintz et al., 2019). A study conducted by Liu et al. (2015) illustrates this flexible relationship. The experimenters asked expert and novice poets to improvise and then revise poems in an fMRI scanning session. They reported deactivation and dissociation of FPN areas during the generation phase, but increased activation and collaboration with other areas during the evaluation phase.5
Such findings indicate that rather than dependence on top-down or bottom-up attention, creative cognition is characterized by leveraging both attentional processes to generate, modify, and evaluate ideas and products (Beaty et al., 2016). The flexible relationship between top-down and bottom-up attention is reflected in recent models of creative cognition. For example, the Model of Creativity and Attention (MOCA) suggests that different configurations of top-down and bottom-up attention might be at work depending on different components and stages of creativity (Vartanian, 2009; Zabelina, 2018; Zabelina et al., 2019; Zabelina & Robinson, 2010). Notably, while a flexible relation between top-down and bottom-up attention is conducive to most creative activities, the extent of their contribution depends on the type, context, and constraints of each creative activity (Chrysikou et al., 2014; Pinho et al., 2015; Zabelina & Ganis, 2018).
This flexible view offers a more holistic and consistent account of attentional mechanisms of creative cognition. However, even this flexible view leaves some significant components of creative cognition unexplained. As we will see in the following section, spontaneous self-generated thoughts, imagination, and mind-wandering make integral contributions to idea generation. This internal and self-generated content does not exactly align with the standard view of bottom-up attention since its source is not the surrounding world.6 Similarly, such content does not perfectly fit the top-down definition since it might occur involuntarily and in a task-irrelevant manner that can distract from the task-at-hand. Thus, the top-down/bottom-up taxonomy falls short of fully explaining the role of mental contents in creative cognition. Below, we review a different taxonomy of attention, internal vs. external, that does a better job of capturing such content.
3.2 Internal versus external attention
Internal and external attention is a newer taxonomy coined by Chun et al. (2011). This taxonomy was proposed partly to counter the view of attention as a unitary construct or mechanism, instead asserting that attention is “a characteristic and property of multiple perceptual and cognitive control mechanisms” (Chun et al., 2011, p. 74). Instead of focusing on the source of input, Chun, Golomb, and Turk-Browne are concerned with the targets of attention. The targets of external attention are externally-generated (e.g., sensory stimuli), whereas the targets of internal attention are internally-generated (e.g., memories, Chun et al., 2011, p. 77). Imagine sitting in front of your computer at work: external attention would include focusing on the computer screen, carefully listening to a conversation in the hallway, or enjoying the taste of coffee while sitting at your desk, while internal attention would include focusing on your thoughts, thinking about a talk you attended, trying to remember the name of an author, or deciding where to go for lunch (Chun et al., 2011). Of course, there is substantial overlap between internally and externally-generated mental content—focusing on a talk might be external attention one day and internal attention the next. Along those lines, subsequent work has found internal and external attention to share significant resources: “external and internal attention mostly operates based on a common, central pool of resources” (Verschooren et al., 2019, p. 478).
External attention is mainly supported by visual, somatomotor, and dorsal attention (DAN) brain networks. The somatomotor network is responsible for movements and touch, comprising primary motor, premotor, and primary sensory cortices. DAN allows for deliberate attention to visual stimuli and spatial locations. In contrast, internal attention is supported by the “limbic” network and DMN. The limbic network, including the subgenual anterior cingulate cortex and the amygdala, involves the processing of emotions (Zabelina & Andrews-Hanna, 2016). DMN mainly consists of medial prefrontal cortex, the posterior inferior parietal cortex, and the medial temporal lobe. Together, DMN regions support memory retrieval, imagination, future planning, metacognition, mental inference, and self-reflection (Buckner et al., 2008; Raichle et al., 2001). Both internal and external attention can couple with cognitive control areas to support goal-directed behavior (Dixon et al., 2014; Zabelina & Andrews-Hanna, 2016).
Recent work emphasizes the importance of internal attention for creative cognition based on supporting evidence from behavioral and neuroimaging studies (Beaty et al., 2019; Benedek, 2018; Benedek & Fink, 2019). Below, we review this evidence and argue that internal attention is not sufficient for the fullest expressions of creative cognition. Instead, creativity is best supported by a dynamic interplay between internal and external attention and, as we will argue, flexible cognitive control.
3.2.1 The role of internal attention in creative cognition
Mind-wandering is often characterized by disengagement from the immediate environment, thinking about what is unrelated to the here and now (Schooler et al., 2011; Smallwood et al., 2007). It is thought to result from internal attention but can be spontaneous or deliberate (Zedelius & Schooler, 2020). Due to its disruptive effects on task performance, mind-wandering has been traditionally associated with adverse effects. Studies show that frequent mind-wandering impairs performance on tasks that require sustained engagement with the external world, such as learning, reading, and driving (for a review, see Mooneyham & Schooler, 2013). Despite its negative influence on task performance, mind-wandering has been repeatedly shown to be positively related to creative cognition (Agnoli et al., 2018; Baird et al., 2012; Fox & Christoff, 2018; Gable et al., 2019; Zedelius & Schooler, 2020).
One study reported that a considerable proportion of creative ideas occur during episodes of mind-wandering (Gable et al., 2019). In this study, experimenters observed professional writers and physicists to examine the unfolding of creative ideas over two weeks. Participants were asked what they were doing or thinking when a creative idea occurred and how creative they thought it was. Results indicated that a significant proportion (20%) of reported creative ideas happened during episodes of mind-wandering. In addition, mind-wandering was particularly conducive to creative ideas that occurred after the individual hit an impasse while working on a problem (Gable et al., 2019).7
In another study, Agnoli et al. (2018) asked participants to complete self-report measures on their tendency to engage in deliberate and spontaneous mind-wandering as well as a creative achievement questionnaire. They also instructed participants to develop creative titles for well-known novels and movies (a divergent thinking task). Results suggested that a higher tendency for deliberate mind-wandering was associated with more original ideas. During episodes of mind-wandering, mental content shifts toward unsolved problems, revisiting and exploring them with fewer constraints and from new angles that can lead to novel solutions (Gable et al., 2019).
Internal attention has become a focal point in creativity research due to its contributions to memory retrieval, prospective planning, and mental simulations (Benedek et al., 2016; Benedek, 2018). Furthermore, as we will show later, the coupling of internal attention with cognitive control mechanisms allows for the active generation, manipulation, and evaluation of ideas (Beaty et al., 2019; Dixon et al., 2014; Zabelina & Andrews-Hanna, 2016). Nonetheless, mounting evidence points to possible contributions to creative cognition from external attention.
3.2.2 The role of external attention in creative cognition
Working memory, and increased working memory capacity, is known to play an important role in creative cognition (see, e.g. Gong et al., 2023), since it allows one to hold multiple ideas in a single space, examining ways in which they might be combined. Yet, working memory can be extended externally, allowing us to use our environment in the process of creative cognition (see, e.g., Maglio & Kirsh, 1992). In that vein, we explore the contribution of external attention to creative cognition.
A series of studies reports lower performance in different creative tasks when the external stimulus is masked (Benedek et al., 2011, 2016).8 In a within-subject design, participants performed either a convergent or divergent creative task.9 In half of the trials, the task stimulus was masked after 500 milliseconds (long enough to clearly see the stimulus), while in the other half, the stimulus remained unmasked for the entire duration of task performance. Results showed that performance in both convergent and divergent tasks was significantly higher in the unmasked condition relative to the masked condition. A possible explanation for this finding is that in the unmasked condition, the stimulus can be retrieved externally at any time, freeing up working memory resources. These additional resources can then be used for further higher-order processing and manipulation of mental representations, allowing for greater creativity. The masked condition, in contrast, would be associated with a higher load on working memory, since the stimulus has to be maintained and processed in memory throughout the task (Benedek et al., 2011, 2016). In our view, the masked condition approximates the absence of external attention, since in both cases the agent is missing access to the external stimulus. Thus, this study demonstrates the ways in which external attention might support creative cognition.
Observations of real-life creative performance—and not just tasks confined to laboratory settings—also point to the importance of dynamic interactions between external and internal attention. For example, when working on non-trivial proofs and problems, mathematicians tend to not only focus on the internal contents of the mind, but they do so while constantly interacting with the environment by creating and manipulating diagrams and inscriptions using chalk and blackboard or pen and paper (Johansen & Misfeldt, 2020; Marghetis et al., 2019). We see this facilitation as akin to an extension of working memory, enabling greater creativity.
It is clear in the above cases that external attention is conducive to creative cognition. We view its role as supplementary to internal attention, while nonetheless making significant contributions to the creative process. It does this, in part, by using the body and surrounding environment as extended memory, allowing for greater allocation of working memory resources toward the generation and manipulation of novel mental representations (Ballard et al., 1997; Clark & Chalmers, 1998; Goldin-Meadow et al., 2001). One might look at this through the lens of “material anchors,” often used to describe the role of external representations in cognition. The thought is that humans project abstract concepts to external representations in order to anchor (stabilize) their understanding of such concepts. Anchoring can lighten the load of memory and facilitate further higher-order processing and inference making (Hutchins, 2005; see also Fauconnier & Turner, 2002). Mathematicians’ constant interactions with diagrams and notations while working on non-trivial problems reflect the significance of external representations in creative cognition.
3.3 Combining top-down/bottom-up and internal/external attention for creative cognition
How are the two taxonomies we reviewed here related to each other? One might be tempted to map internal attention to top-down and external attention to bottom-up, since external stimuli are typically responsible for bottom-up attention. However, it is best to think of the two taxonomies as orthogonal. Both internal and external attention can exhibit properties of either bottom-up or top-down attention. For example, external attention can be either task-relevant (i.e. top-down) or stimulus-driven (i.e. bottom-up) so long as its target is external stimuli. Similarly, internal attention can be either top-down or bottom-up. Intrusive thoughts or spontaneous mind-wanderings during which an individual unintentionally thinks about events or memories disconnected from the present time or location would count as bottom-up attention, whereas intentional and directed mind-wandering—for instance to plan future events, or to remember someone’s address—would count as top-down attention (Dixon et al., 2014). There is, thus, no one-to-one map between the bottom-up/top-down and internal/external taxonomies of attention.
Consequently, we suggest that the role of attention in creative cognition can be depicted as a two-dimensional space, with the target of attention (i.e., internal and external) as one dimension and the source of attention (i.e., top-down and bottom-up) as the second dimension. We argue that this two-dimensional space sufficiently accounts for the reported flexibility and diversity of attention across different stages, components, and constraints of creative endeavors (see figure 1).
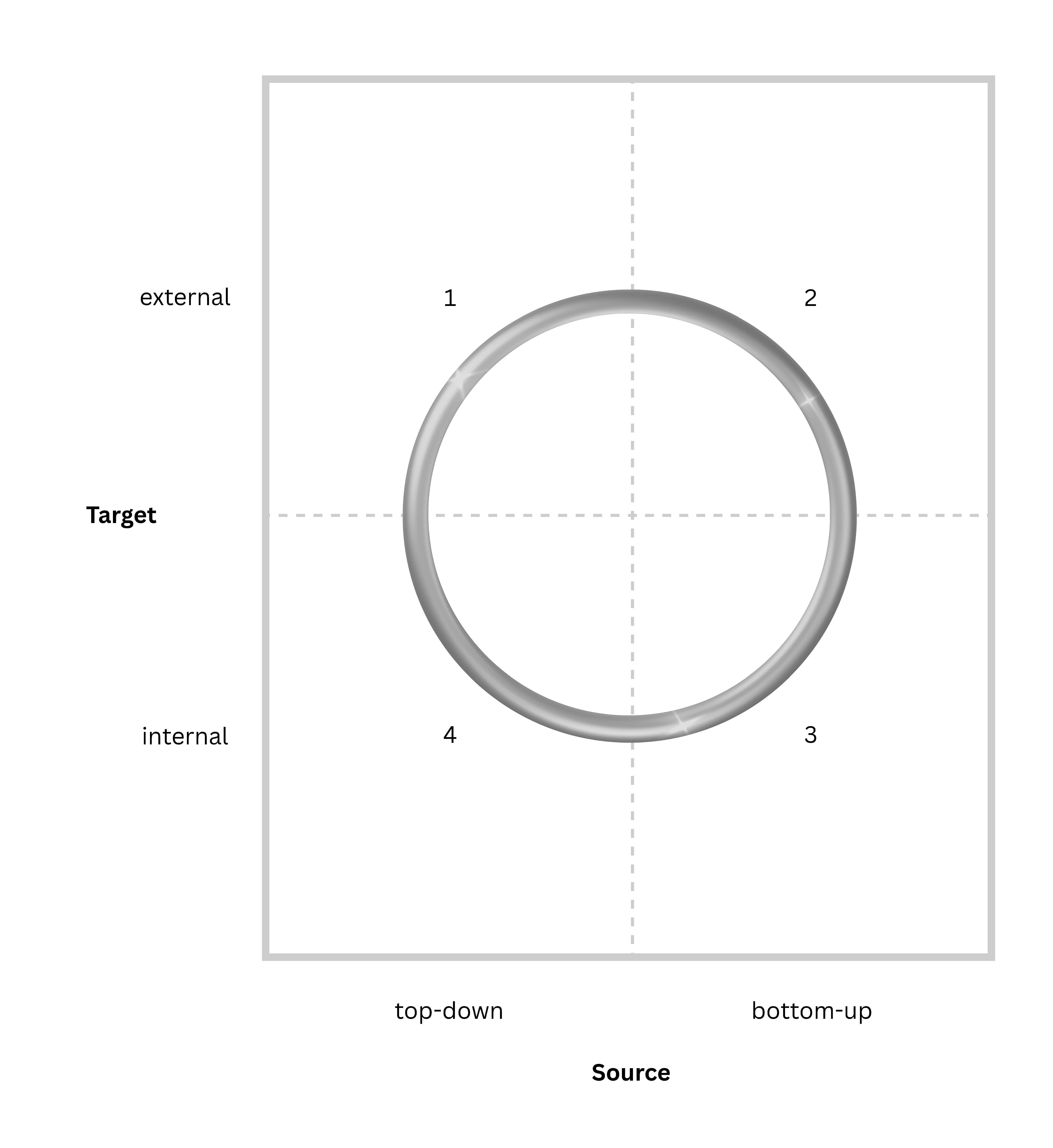
This two-dimensional space is continuous but includes four zones of attention: external and top-down, external and bottom-up, internal and bottom-up, internal and top-down. Zone 1 (external and top-down) captures instances of creative cognition when individuals intentionally pay attention to the external world. For example, in a jazz quartet musicians actively listen to the music made by other group members while also monitoring any feedback from the audience (Dixon et al., 2014). Likewise, when working on a problem, mathematicians actively make inscriptions and interact with them (Marghetis et al., 2019). Artists, too, intentionally pay attention to the physical properties of various media—the canvas, the colors, the clay, etc.—to adapt their creative endeavor to the constraints of each medium.
Zone 2 (external and bottom-up) captures instances of creativity that are triggered in a bottom-up fashion; that is, by the salient physical properties of some object in the world. Accounts of serendipitous innovations and leaky attention (Zabelina et al., 2016) suggest the involvement of this attentional zone. Zone 2 might contribute to creative cognition by adding randomness to the search process. Randomness can reduce bias for prior knowledge and common ideas which can in turn increase the probability of exploring a wider area of the search space (Kirsh, 2014). For example, during jazz improvisation, musicians might produce novel and surprising notes inspired by perceived bottom-up cues from the audience or notes generated by other group members (Ramalho & Ganascia, 1994). A famous case of this is the serendipitous discovery of penicillin by Fleming that occurred by his noticing an unusual pattern of bacterial growth in a petri dish (Gaynes, 2017).
Zone 3 (internal and bottom-up) captures spontaneous internal thoughts that might distract from the task-at-hand but contribute to an overall creative goal. An example is Poincaré’s account of the sudden discovery of a solution to a non-trivial mathematical problem while stepping onto a bus during vacation (Poincaré, 2015). Likewise, Kekulé’s account of his major discovery of the hexagonal structure of the benzene molecule indicates that the idea came to him while daydreaming (see Rothenberg, 1995)—or what we refer to as episodes of spontaneous mind-wandering. Similar to Zone 2, Zone 3 might also contribute to creativity through increased randomness, and thus, a more explorative search. But unlike Zone 2, the randomness in Zone 3 might primarily originate from internal thoughts, not salient stimuli in the immediate surrounding world.
Zone 4 (internal and top-down) captures instances of creative cognition that rely on guided memory retrieval and maintenance of internal trains of thought through inhibition of irrelevant internal and external stimuli. Lab-based creativity tests such as AUT and RAT mostly involve this attentional zone; such tasks require minimal interaction with the outside world and are characterized by goal-directed memory retrieval and mental simulations.
While some creative tasks can be located in one of the four zones (e.g., AUT), most creative endeavors require interactions between multiple attentional zones encompassing internal, external, bottom-up and top-down forms of attention. Take our earlier example of the musical improvisation of a jazz quartet. At some stages of the process, internally-oriented attention might be enough to spontaneously generate musical ideas, which are then translated into motor activities resulting in external sounds. Yet, at most stages the musicians will also need cognitive control to make dynamic shifts between internal and external attention in order to optimize the creative process. The cooperation and coordination of top-down and bottom-up, internal and external attention allows musicians to be flexible and adapt themselves to unexpected changes in the musical patterns. Jazz musicians retrieve prior knowledge of music and generate new musical patterns while physically interacting with an instrument and monitoring other band members, the overall rhythmic patterns, and feedback from the audience (see Dixon et al., 2014; Loui & Guetta, 2019; Ramalho & Ganascia, 1994).
The cooperation and coordination across forms of attention is also evident when we zoom in on accounts of major discoveries by Poincaré, Fleming, and Kekulé, mentioned above. At first glance, these discoveries might seem solely reliant on attentional Zones 2 and 3. Nevertheless, in all three scenarios, the scientists had already started familiarizing themselves with the constraints of each problem and its corresponding solution space. What’s more, these accounts indicate that after finding the solution, the scientists deliberately tested the solution to make sure of its viability (Gaynes, 2017; Poincaré, 2015; Rothenberg, 1995). That is, the formation of the solution was both preceded and followed by attentional Zones 1 and 4. In other words, the creative process, from start to end, includes dynamic configurations of internal, external, top-down, and bottom-up attention. Such dynamic attentional mechanisms of creative cognition are also reflected in recent neuroimaging studies of creativity. We will review some of these studies in the next section.
3.3.1 Dynamic attentional mechanisms of creative cognition as evidenced by neuroimaging studies
As mentioned earlier, internal and external attention can both draw on cognitive control to fulfill goal-directed behavior. In addition, internal and external attention can communicate and interact with each other through FPN and SN (Zabelina & Andrews-Hanna, 2016). SN mainly consists of the anterior insula and dorsal anterior cingulate cortex (Menon & Uddin, 2010; Uddin, 2017). The core function of this network is to identify salient stimuli (internal or external), mark them for additional processing, and guide target brain regions to generate the appropriate behavioral response.
Neuroimaging studies report a positive coupling between areas related to internal attention (DMN) and those related to cognitive control (FPN) during creative performances (Beaty et al., 2018; De Pisapia et al., 2016; Ellamil et al., 2012; Shi et al., 2018). Consistent reports of this positive coupling suggest that although internal attention supports creativity by facilitating memory retrieval and mental simulations, it is insufficient. Any retrieved memory contents and mental representations will require further manipulation to satisfy task constraints and stay in line with the creative goal. Such goal-directed behavior is best supported by cognitive control mechanisms and their realizers (i.e., FPN). Further, goal-directed internal attention needs to be maintained and shielded from distracting stimuli (both internal and external) over time; this function, too, is effectively fulfilled by cognitive control. Additionally, depending on the type and constraints of the creative task, individuals might need to actively interact with the surrounding world, get inspired by, and/or manipulate relevant external stimuli.
Thus, creative cognition seems to maximally benefit from dynamic interactions between internal and external attention as well as cognitive control, such as top-down attention. This is evident from a study during which fMRI data were recorded from 163 participants while performing a divergent thinking task (i.e., AUT) and a control task (asking participants to think of typical characteristics of everyday objects, Beaty et al., 2018). The data were then used to identify a whole-brain network associated with high-creative ability, which indicated that highly creative individuals can simultaneously engage three large-scale brain networks—DMN, FPN, and SN (Beaty et al., 2018). This is in notable contrast to the observed anti-correlation of DMN and FPN in other, non-creative tasks (see, e.g., Fox et al., 2005).
A similar pattern of simultaneous engagement of three large-scale brain networks is reported for instances of mind-wandering. Christoff et al. (2009) conducted an fMRI study to examine the neural mechanisms of mind-wandering. They used an experience sampling method to intermittently probe participants about their current mental state as they performed a simple go/no-go task. Results indicated that DMN, FPN, and SN regions were all recruited during mind-wandering episodes (for similar results, see Fox et al., 2015; Fox & Christoff, 2018).
Together, the neuroimaging studies of mind-wandering and creative task performance attest to dynamic attentional mechanisms underlying creative cognition. Beaty et al. (2018, p. 1090) explain this as follows:
The default network contributes to the generation of ideas via flexible and spontaneous combinatory mechanisms involved in memory retrieval and mental simulation. The salience network, in turn, functions to identify candidate ideas—potentially useful information generated via the default network—and forward such information to frontoparietal executive systems for high-order processing (e.g., idea evaluation, elaboration, or revision).
Effective communication across multiple attentional forms is reflected in the coupling of DMN and FPN through SN, which upon detecting a salient event, facilitates rapid access to relevant memory, attention, and motor resources (Menon & Uddin, 2010).
4 Discussion
Creative cognition is characterized as a biphasic phenomenon, typically moving from idea generation to idea evaluation. Both phases require selection in one form or another, whether to direct the process of generation or to support the process of evaluation. Thus, examining attentional mechanisms is an excellent way to gain insight into creative cognition. We did this through the lens of two major taxonomies: top-down versus bottom-up attention, and internal versus external attention. Previous research focused on the presence or absence of attention, typically through a single form of attention. We discussed, for example, studies that report either reduced top-down attention or increased internal attention as conducive to creative cognition. However, we find that a dynamic relationship between different forms of attention is most beneficial for creative cognition. Various configurations of top-down and bottom-up, internal and external attention can benefit creativity; some configurations are more useful during idea generation (Zone 2 and Zone 3), whereas others are more useful during idea evaluation (Zone 1 and Zone 4).
The dynamic interplay between different forms of attention is reflected in the neural underpinnings of creative cognition, and especially the close interaction between DMN, FPN, and SN. DMN and FPN are typically anti-correlated, with DMN being more active during resting states and FPN more active during tasks that rely on interactions with external stimuli. During resting states, one tends to engage with internal contents (e.g., memories) without exerting top-down control. Hence, FPN shows reduced activation, while DMN shows increased activation. In contrast, during tasks that rely on interactions with external stimuli, mind-wandering can distract and impair performance. Top-down control is needed to keep one engaged with the task and inhibit unwanted thoughts (such as memories). Hence, in this scenario, FPN shows increased activation, while DMN shows reduced activation.
Creative cognition, however, requires that we think beyond the typical opposition between task engagement and resting state. Creative cognition requires cognitive control mechanisms, such as top-down attention, to maintain, manipulate, and assess ideas that are generated according to the constraints of the creative task. Yet, it also requires that new ideas are generated “bottom-up,” which can occur through both internal (e.g., memory retrieval) and external (e.g., sensing) mechanisms. This flexibility and cooperation—across DMN, FPN, and sensory cortices—is facilitated by SN through a moment-by-moment identification of salient events (internal or external) and allocation of relevant memory, attentional, and motor resources to elicit an appropriate behavioral response. This is in contrast with non-creative tasks, which can be harmed by insufficient exclusion of other brain networks and stimuli.
The coupling between DMN and FPN has also been reported in phenomena often associated with enhanced creativity, including mind-wandering (see the previous section), altered states of consciousness induced by hallucinogen consumption (Palhano-Fontes et al., 2015), and meditation (Hasenkamp & Barsalou, 2012; Josipovic et al., 2012).
To further explain the contributions of DMN/FPN coupling to creative cognition, some draw an analogy to simulated annealing (Carhart-Harris & Friston, 2019; Girn et al., 2020; Gopnik et al., 2017). Annealing is a metallurgy term that refers to the process of repeatedly heating a metal and then slowly cooling it down, resulting in a more robust structure. Similarly, in computer science, the optimal solution is reached when the system starts with a broader (i.e., high-temperature) search and gradually moves to a narrower (i.e., low-temperature) search. A heated system is more flexible in its movements across the possible solutions, which enables it to escape local minima and continue the search until the optimal solution is reached (Kirkpatrick et al., 1983; Metropolis et al., 1953). Likewise, the initial enhanced coupling between DMN and SN reflects a heated, explorative, and flexible search suitable for idea generation in creative cognition. The subsequent coupling between DMN and FPN through SN reflects the gradual cooling of the system, which allows for assessment, revision, elaboration, and selection of candidate ideas.
5 Summary and conclusion
Creative cognition is a complex phenomenon emerging from interactions across common cognitive processes, such as memory, executive functions, and attention. The present paper reviewed attentional mechanisms of creative cognition with an emphasis on two major taxonomies: top-down/ bottom-up and internal/ external attention. These taxonomies can be conceived as orthogonal: the division between top-down and bottom-up attention is based on the source of attentional input, while the division between internal and external attention is based on the target of attention. We argued that engagement across the taxonomies best captures the complex and flexible nature of creative cognition. Creativity does not solely depend on top-down or bottom-up processes; neither does it solely depend on internal or external attention. A dynamic interplay between these different forms of attention is required to best enable novel ideas' generation, evaluation, and elaboration. We support these claims with research showing that dynamic interaction between types of attention in each taxonomy is conducive to creativity, as well as supporting research on the neural basis of creative cognition. However, we also found that the extent of contribution from each type hinges on the context and constraints of the creative activity. Thus, determining the most conducive contributions of each type will require further research.
References

